 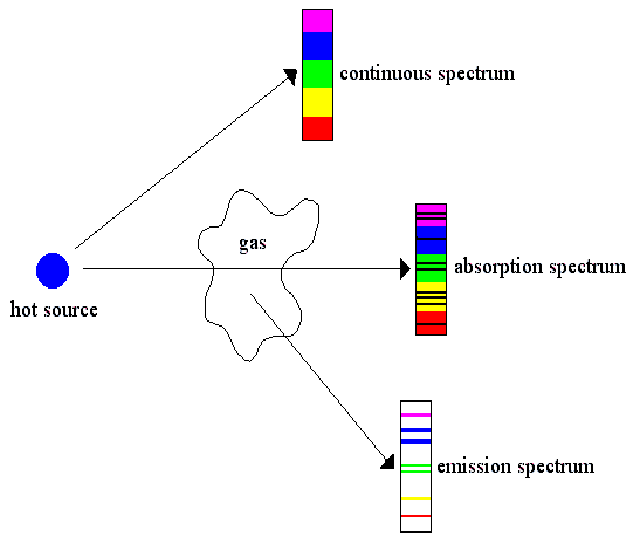
The absorption spectrum can be defined as a spectrum of the spectral lines resulting from the absorption of electromagnetic radiation. Therefore, given the two types of spectra, absorption spectra correspond to an increase in kinetic energy (of electrons), whereas emission spectra correspond to a decrease in kinetic energy (of electrons). The reverse is true in the case of emission spectra light emitted by atoms is produced as electrons drop down from a higher energy level to a lower one. Thus, the absorption spectrum is obtained when electrons are made to jump from a lower energy level to higher levels. This is because light can be absorbed only when it makes an electron jump to a higher energy level. Where, E – the energy of the photon (Jmol-1)Ībsorption and emission spectra are the manifestations of atomic transitions. The energy of the given Photon is E = hc / λ When the element is cold, there will be no absorption of radiation, and so one gets an emission spectrum. 
The beam of light is passed through the vapor of an element. The light source, such as a light bulb or fluorescent tube, is used to produce electromagnetic radiation of all wavelengths. The highlight of absorption spectra is that it shows dark lines on the spectrum. This means that there must be an energy source like burning a substance or heating it intensely to generate emission spectra. Emission spectra will consist of bright lines formed when the atoms jump from a higher shell to a lower one while they emit energy during the process. The first one has a prominent feature that is dark lines produced due to absorption between two bright lines. These are emission and absorption spectra. Spectra may be divided into two classes according to the manner in which light interacts with the atoms of a gas or a liquid. This Movement Causes the absorption and emission Spectrum. When energy is given to an atom externally, it makes the electrons move from one shell to another. The shell which is closer to the nucleus has the lowest Energy compared to others. According to modern atomic theory, these electrons are arranged in energy levels, specifically called shells or orbitals, where their energies are quantized. An atom in an element has a central core called a nucleus and a cloud of electrons around the nucleus. These electrons are positioned in specific energy levels called shells or orbitals that surround the nucleus in a pattern that corresponds to their energies. An element is composed of a specific number of protons, the nuclear core of an atom, and its properties are determined by the number of electrons orbiting the nucleus. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
